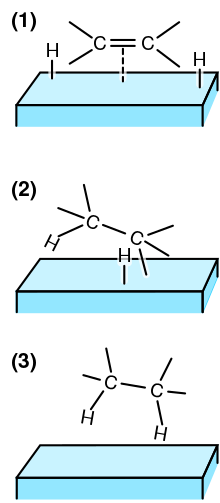
29.8: A Catalyst Affects the Mechanism and Activation Energy - Chemistry LibreTexts
Homogeneous catalysis refers to reactions in which the catalyst is in solution with at least one of the reactants whereas heterogeneous catalysis refers to reactions in which the catalyst is present …
Homogeneous catalysis refers to reactions in which the catalyst is in solution with at least one of the reactants whereas heterogeneous catalysis refers to reactions in which the catalyst is present in a different phase, usually as a solid, than the reactants.
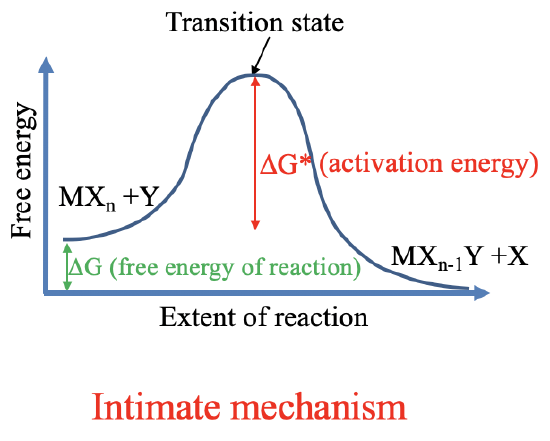
9.1: Substitution Reactions and their Mechanisms - Chemistry LibreTexts
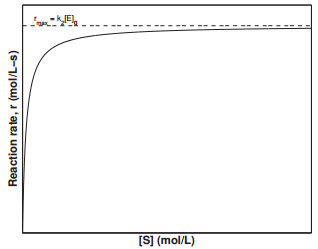
29.9: The Michaelis-Menten Mechanism for Enzyme Catalysis - Chemistry LibreTexts

Student Experiment: Rates of Chemical Reactions

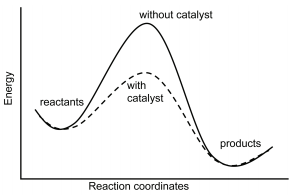
6.10: Energies, Kinetics, and Catalysts - Chemistry LibreTexts

487928109-Physical-Chemistry-McQuarrie-and-Simon-Full.pdf
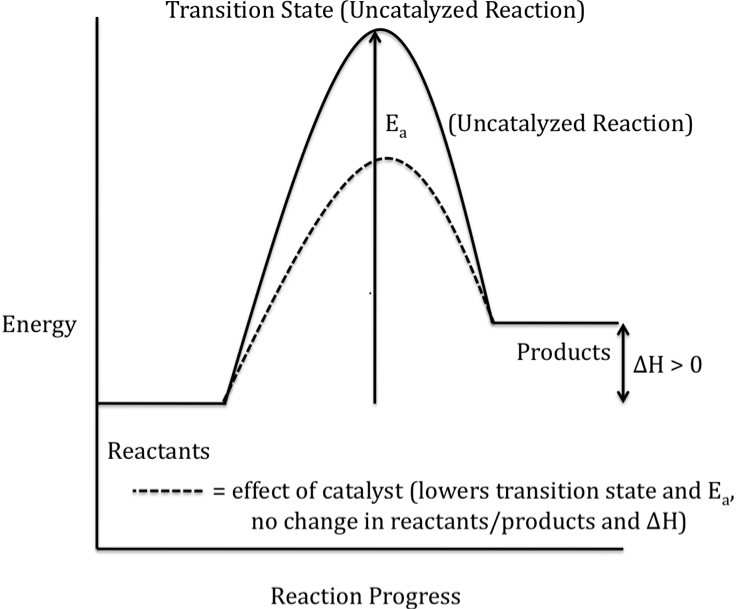
How is the energy level of the products affected by the presence of a catalyst?

487928109-Physical-Chemistry-McQuarrie-and-Simon-Full.pdf

18.7 Catalysis – Chemistry Fundamentals
How is activation energy affected by the catalyst? - Quora
How is activation energy affected by the catalyst? - Quora

Ideal Gas Law Lab
12.7: Catalysis - Chemistry LibreTexts